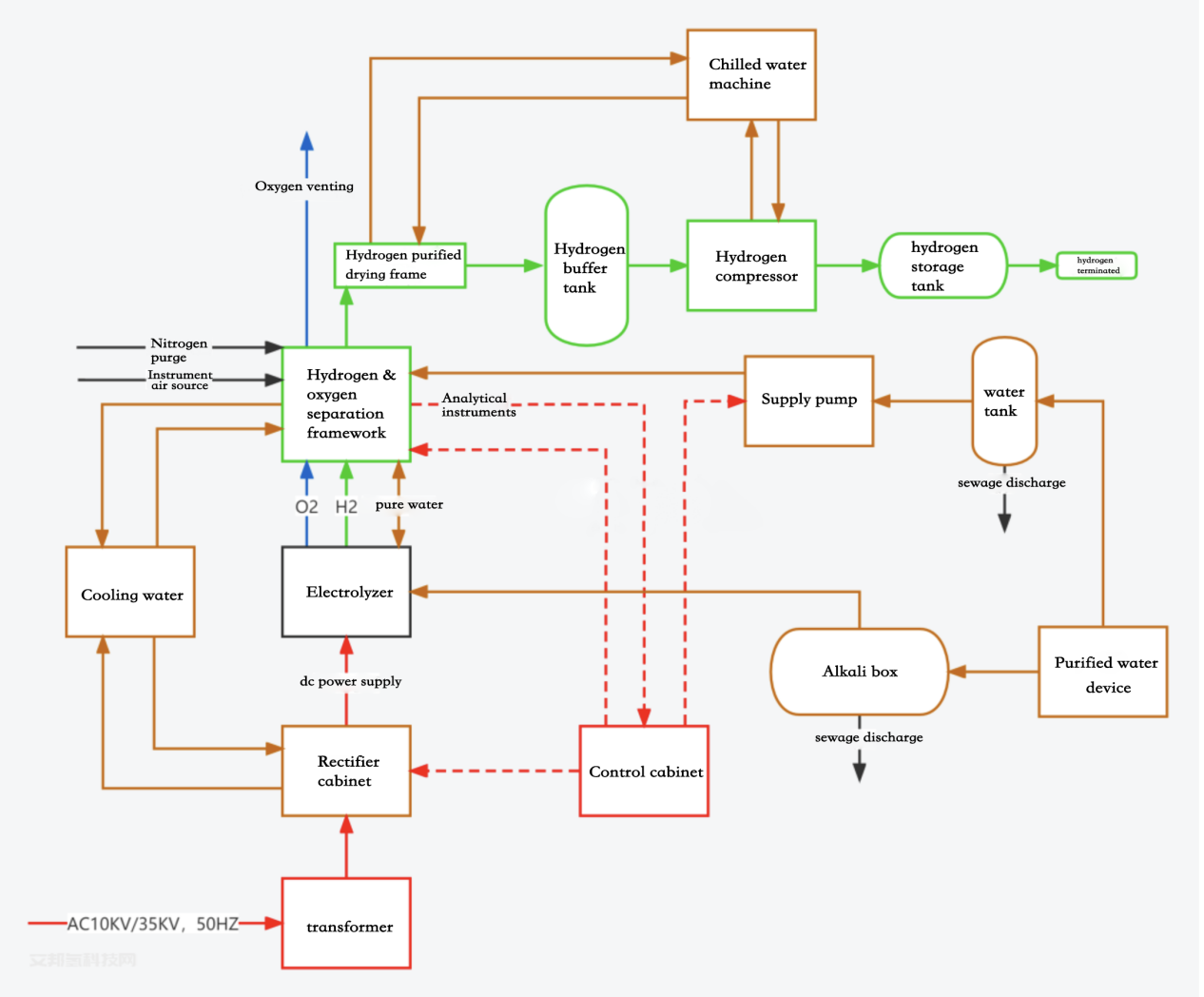
Sashen samar da hydrogen na electrolysis ya haɗa da cikakken kayan aikin samar da hydrogen na electrolysis na ruwa. Babban kayan aikin shine:
1. Elektroliza
2. Na'urar raba ruwa mai iskar gas
3. Tsarin busarwa da tsarkakewa
4. Sashen wutar lantarki ya haɗa da: na'urar canza wutar lantarki, kabad ɗin gyarawa, kabad ɗin sarrafa shirye-shiryen PLC, kabad ɗin kayan aiki, kabad ɗin rarraba wutar lantarki, kwamfuta mai masauki, da sauransu.
5. Tsarin taimako ya ƙunshi: tankin alkali, tankin ruwa na kayan masarufi, famfon samar da ruwa, kwalbar nitrogen/sandar bas, da sauransu.
6. Tsarin taimako na gaba ɗaya ya haɗa da: injin ruwa mai tsarki, hasumiyar ruwan sanyaya, injin sanyaya, na'urar sanyaya iska, da sauransu.
A cikin sashin samar da hydrogen na lantarki, ruwa yana narkewa zuwa wani ɓangare na hydrogen da kashi 1/2 na oxygen a cikin electrolyzer ƙarƙashin aikin wutar lantarki kai tsaye. Ana aika hydrogen da oxygen da aka samar zuwa ga mai raba gas-liquid tare da electrolyte don rabuwa. Ana sanyaya hydrogen da oxygen ta hanyar sanyaya hydrogen da oxygen, kuma mai kama drop catcher yana kamawa da cire ruwa, sannan ana aika shi ƙarƙashin ikon tsarin sarrafawa; electrolyte yana wucewa ta hanyar hydrogen, matatar alkali ta oxygen, hydrogen, matatar alkali ta oxygen, da sauransu a ƙarƙashin aikin famfon zagayawar jini. mai sanyaya ruwa sannan a mayar da shi zuwa electrolyzer don ci gaba da electrolysis.
Ana daidaita matsin lambar tsarin ta hanyar tsarin sarrafa matsin lamba da tsarin sarrafa matsin lamba daban-daban don biyan buƙatun ayyuka da ajiya na gaba.
Hydrogen da aka samar ta hanyar electrolysis na ruwa yana da fa'idodin tsarki mai yawa da ƙarancin ƙazanta. Yawanci, ƙazanta a cikin hydrogen da aka samar ta hanyar electrolysis na ruwa kawai oxygen ne da ruwa, kuma babu wasu abubuwan haɗin gwiwa (waɗanda za su iya guje wa guba daga wasu abubuwan kara kuzari), wanda ke ba da sauƙi don samar da hydrogen mai tsafta. , bayan tsarkakewa, iskar da aka samar za ta iya isa ga alamun iskar gas ta masana'antu ta lantarki.
Hydrogen da na'urar samar da hydrogen ke samarwa ta ratsa ta cikin tankin buffer don daidaita matsin lambar aiki na tsarin da kuma cire ruwa kyauta a cikin hydrogen.
Bayan hydrogen ya shiga na'urar tsarkake hydrogen, hydrogen da ruwa ke samarwa za a ƙara tsarkake shi, kuma ana cire iskar oxygen, ruwa da sauran ƙazanta a cikin hydrogen ta amfani da ƙa'idodin catalytic reaction da molecular sieve adsorption.
Kayan aikin na iya saita tsarin daidaitawa ta atomatik don samar da hydrogen bisa ga ainihin yanayin. Canje-canje a cikin nauyin iskar gas zai haifar da canzawa a cikin matsin lamba na tankin ajiyar hydrogen. Mai watsa matsin lamba da aka sanya a kan tankin ajiya zai fitar da siginar 4-20mA kuma ya aika ta zuwa PLC kuma Bayan kwatanta ƙimar saitin asali da yin canjin juyi da lissafin PID, ana fitar da siginar 20 ~ 4mA kuma a aika zuwa kabad ɗin gyara don daidaita girman wutar lantarki ta lantarki, don haka cimma manufar daidaita samar da hydrogen ta atomatik bisa ga canje-canje a cikin nauyin hydrogen.

Kayan aikin samar da hydrogen na ruwa alkaline electrolysis galibi sun haɗa da tsarin masu zuwa:
(1) Tsarin ruwa na kayan ƙasa

Abu ɗaya tilo da ke amsawa a cikin tsarin samar da hydrogen ta hanyar lantarki shine ruwa (H2O), wanda ke buƙatar a ci gaba da cika shi da ruwa mai ɗanɗano ta hanyar famfon sake cika ruwa. Matsayin sake cika ruwa yana kan mai raba hydrogen ko oxygen. Bugu da ƙari, dole ne a cire ƙaramin adadin hydrogen da oxygen lokacin da ya bar tsarin. Yawan ruwan da ƙananan kayan aiki ke sha shine 1L/Nm³H2, kuma na manyan kayan aiki za a iya rage shi zuwa 0.9L/Nm³H2. Tsarin yana ci gaba da cike ruwan danye. Ta hanyar sake cika ruwa, ana iya kiyaye daidaiton matakin ruwa na alkaline da yawan alkali, kuma ana iya sake cika ruwan da ke cikin lokaci don kiyaye yawan ruwan da ke cikin ruwa.
2) Tsarin gyara na'urar canza wutar lantarki
Wannan tsarin ya ƙunshi na'urori guda biyu: na'urar canza wutar lantarki da kuma kabad ɗin gyara wutar lantarki. Babban aikinsa shine canza wutar lantarki ta AC mai ƙarfin 10/35KV da mai shi na gaba ke bayarwa zuwa wutar lantarki ta DC da na'urar lantarki ke buƙata, sannan a samar da wutar lantarki ta DC ga na'urar lantarki. Ana amfani da wani ɓangare na wutar lantarki da ake bayarwa don lalata ruwa kai tsaye. Kwayoyin halittar sune hydrogen da oxygen, ɗayan ɓangaren kuma yana samar da zafi, wanda mai sanyaya ruwan leda ke fitarwa ta hanyar ruwan sanyaya.
Yawancin na'urorin canza wutar lantarki na nau'in mai ne. Idan aka sanya su a cikin gida ko a cikin akwati, ana iya amfani da na'urorin canza wutar lantarki na nau'in busasshe. Na'urorin canza wutar lantarki da ake amfani da su a cikin kayan aikin samar da hydrogen na ruwa na lantarki na musamman ne kuma suna buƙatar a daidaita su bisa ga bayanan kowace na'urar lantarki, don haka kayan aiki ne na musamman.

(3) tsarin kabad ɗin rarraba wutar lantarki
Ana amfani da kabad ɗin rarraba wutar lantarki galibi don samar da 400V ko kuma wanda aka fi sani da kayan aikin 380V ga sassa daban-daban tare da injina a cikin tsarin rabuwa da tsarkakewar hydrogen da oxygen a bayan kayan aikin samar da hydrogen na ruwa mai amfani da lantarki. Kayan aikin sun haɗa da zagayawar alkali a cikin tsarin rabuwar hydrogen da oxygen. Famfuna, famfunan sake cika ruwa a cikin tsarin taimako; wayoyi masu dumama a cikin tsarin bushewa da tsarkakewa, da tsarin taimako da tsarin gaba ɗaya ke buƙata, kamar injinan ruwa mai tsabta, masu sanyaya iska, masu sanyaya hasumiya, da masu sanyaya hydrogen na baya, injunan hydrogenation da sauran kayan aiki. Samar da wutar lantarki kuma ya haɗa da samar da wutar lantarki don haske, sa ido da sauran tsarin dukkan tashar.
(4) tsarin sarrafawa
Tsarin sarrafawa yana aiwatar da sarrafa PLC ta atomatik. PLC gabaɗaya yana amfani da Siemens 1200 ko 1500. An sanye shi da allon taɓawa na hulɗar ɗan adam da kwamfuta, kuma ana iya ganin aikin da sigogi na kowane tsarin kayan aiki da kuma nuna dabaru na sarrafawa akan allon taɓawa.
5) Tsarin zagayawar Alkali
Wannan tsarin ya ƙunshi manyan kayan aiki masu zuwa:
Mai raba hydrogen da iskar oxygen - famfon zagayawar alkali - bawul - matatar alkali - na'urar lantarki
Babban tsari shine: ruwan alkali da aka gauraya da hydrogen da oxygen a cikin mai raba hydrogen da oxygen an raba shi ta hanyar mai raba gas-liquid sannan ya koma famfon zagayawa na ruwa na alkali. A nan an haɗa mai raba hydrogen da mai raba oxygen, kuma famfon zagayawa na ruwa na alkali zai sake dawowa. Ruwan alkali yana zagayawa zuwa bawul da matatar ruwa ta alkali a ƙarshen baya. Bayan matatar ta tace manyan ƙazanta, ruwan alkali yana zagayawa zuwa cikin electrolyzer.
(6) Tsarin Haydaroji
Ana samar da hydrogen daga gefen electrode na cathode kuma yana isa ga mai raba tare da tsarin zagayawar ruwa na alkali. A cikin mai raba, saboda hydrogen ɗin kansa yana da sauƙi, zai rabu da ruwan alkali ta halitta ya isa saman sashin mai raba, sannan ya ratsa ta bututun don ƙarin rabuwa da sanyaya. Bayan sanyaya ruwa, mai kama digo yana kama digo kuma ya kai tsarkin kusan kashi 99%, wanda ya isa ga tsarin busarwa da tsarkakewa na baya.
Fitowa: Fitowar hydrogen galibi ana amfani da shi ne don fitarwa yayin farawa da rufewa, rashin aiki na yau da kullun ko rashin tsafta, da kuma fitar da lahani.
(7) Tsarin iskar oxygen
Hanya don samun iskar oxygen tana kama da ta hydrogen, amma a cikin wani mai rabawa daban.
Korar mutane: A halin yanzu, yawancin ayyukan iskar oxygen ana magance su ta hanyar kwashe mutane.
Amfani: Darajar amfani da iskar oxygen yana da ma'ana kawai a cikin ayyuka na musamman, kamar wasu yanayi na aikace-aikace waɗanda za su iya amfani da hydrogen da oxygen mai tsafta, kamar masana'antun fiber na gani. Akwai kuma wasu manyan ayyuka waɗanda suka tanadi sarari don amfani da iskar oxygen. Yanayin aikace-aikacen baya sune samar da iskar oxygen mai ruwa bayan bushewa da tsarkakewa, ko amfani da iskar oxygen ta likitanci ta hanyar tsarin watsawa. Duk da haka, har yanzu ba a tantance ingantaccen waɗannan yanayi na amfani ba. Ƙarin tabbaci.
(8) tsarin sanyaya ruwa
Tsarin electrolysis na ruwa martani ne na endothermic. Dole ne a samar da makamashin lantarki ta hanyar samar da hydrogen. Duk da haka, makamashin lantarki da tsarin electrolysis na ruwa ke cinyewa ya wuce shaƙar zafi na ka'idar amsawar electrolysis na ruwa. Wato, wani ɓangare na wutar lantarki da electrolyzer ke amfani da shi ana mayar da shi zafi. Wannan ɓangaren Ana amfani da zafi galibi don dumama tsarin zagayawar alkali a farko, don haka zafin ruwan alkaline ya tashi zuwa kewayon zafin jiki na 90±5°C da kayan aikin ke buƙata. Idan electrolyzer ya ci gaba da aiki bayan ya kai yanayin zafi da aka ƙayyade, ana buƙatar amfani da zafi da aka samar. Ana fitar da ruwan sanyi don kiyaye yanayin zafi na al'ada na yankin amsawar electrolysis. Babban zafin jiki a yankin amsawar electrolysis na iya rage yawan amfani da makamashi, amma idan zafin ya yi yawa, za a lalata membrane na ɗakin electrolysis, wanda kuma zai yi illa ga aiki na dogon lokaci na kayan aiki.
Wannan na'urar tana buƙatar a kiyaye zafin aiki a ƙasa da digiri 95 na Celsius. Bugu da ƙari, dole ne a sanyaya kuma a cire danshi daga hydrogen da oxygen da aka samar, kuma na'urar gyara silicon mai sanyaya ruwa tana da bututun sanyaya da ake buƙata.
Jikin famfo na manyan kayan aiki kuma yana buƙatar shigar da ruwan sanyaya.
(9) Tsarin cika sinadarin nitrogen da tsarkake sinadarin nitrogen
Kafin a gyara na'urar da kuma sarrafa ta, dole ne a cika tsarin da nitrogen don gwajin matsewar iska. Kafin a fara aiki da ita yadda ya kamata, ana kuma buƙatar a tsaftace matakin iskar gas na tsarin da nitrogen don tabbatar da cewa iskar da ke cikin sararin iskar gas a ɓangarorin biyu na hydrogen da oxygen ba ta da nisa da kewayon da ke iya kamawa da wuta.
Bayan an kashe kayan aikin, tsarin sarrafawa zai ci gaba da matsin lamba ta atomatik kuma ya riƙe wani adadin hydrogen da oxygen a cikin tsarin. Idan har yanzu ana samun matsin lamba lokacin da aka kunna kayan aikin, babu buƙatar yin tsaftacewa. Duk da haka, idan an cire duk matsin lambar, za a buƙaci a sake tsaftace shi. Aikin tsarkake nitrogen.
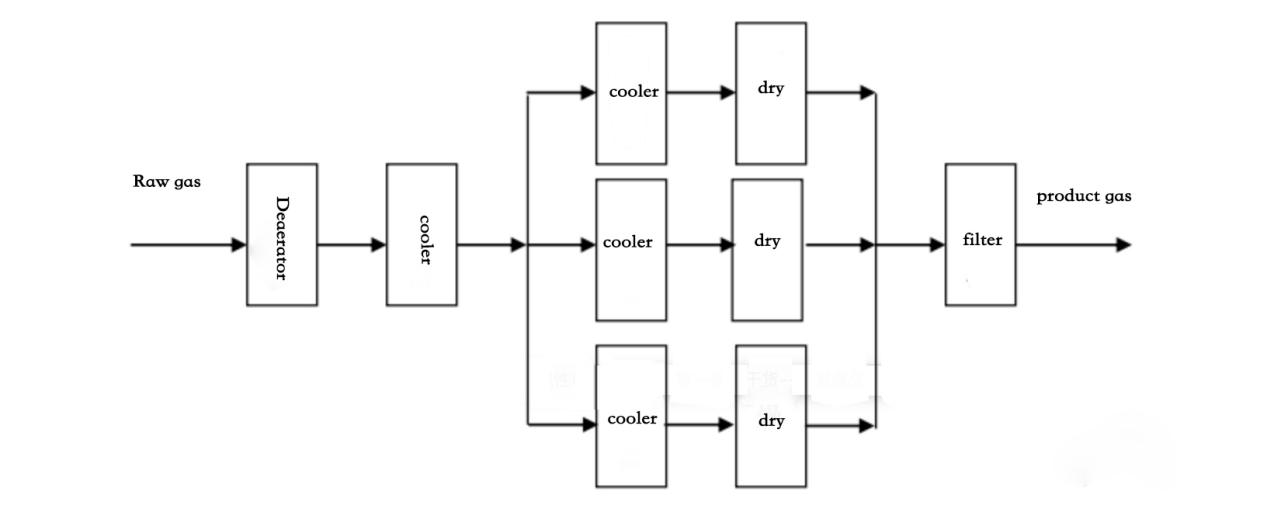
(10) Tsarin busar da hydrogen (tsarkakewa) (zaɓi ne)
Ana cire sinadarin hydrogen da aka samar daga ruwa ta hanyar na'urar busar da shi a layi daya, sannan a karshe a goge shi da matatar bututun nickel mai siminti don samun busasshiyar hydrogen. (Bisa ga buƙatun mai amfani na hydrogen na samfurin, tsarin na iya ƙara na'urar tsarkakewa, kuma tsarkakewar tana amfani da deoxidation na palladium-platinum bimetallic catalytic).
Ana aika sinadarin hydrogen da na'urar samar da hydrogen ta hanyar lantarki ta ruwa zuwa na'urar tsarkake hydrogen ta cikin tankin buffer.
Hydrogen ya fara ratsawa ta hasumiyar deoxygenation. A ƙarƙashin aikin mai kara kuzari, iskar oxygen da ke cikin hydrogen yana amsawa da hydrogen don samar da ruwa.
Tsarin amsawa: 2H2+O2 2H2O.
Sannan, hydrogen ɗin yana ratsa ta cikin na'urar sanyaya iskar hydrogen (wanda ke sanyaya iskar don tarawa tururin ruwa a cikin iskar don samar da ruwa, kuma ruwan da aka tattara ta atomatik yana fita daga tsarin ta hanyar mai tattara ruwa) sannan ya shiga hasumiyar shaye-shaye.
Lokacin Saƙo: Mayu-14-2024




