A duniya, komai yana da nasa fa'idodi da rashin amfani. Ci gaban al'umma da kuma inganta rayuwar mutane ba makawa yana haifar da gurɓatar muhalli. Ruwan shara yana ɗaya daga cikin irin waɗannan matsalolin. Tare da saurin ci gaban masana'antu kamar su sinadarai masu amfani da mai, yadi, yin takarda, magungunan kashe kwari, magunguna, ƙarfe, da samar da abinci, fitar da ruwan shara ya ƙaru sosai a duk duniya. Bugu da ƙari, ruwan shara sau da yawa yana ɗauke da yawan guba, yawan gishiri, da kuma abubuwan da ke ɗauke da launi mai yawa, wanda hakan ke sa ya zama da wahala a lalata shi da kuma magance shi, wanda ke haifar da gurɓatar ruwa mai tsanani.
Domin magance yawan ruwan sharar masana'antu da ake samarwa kowace rana, mutane sun yi amfani da hanyoyi daban-daban, inda suka haɗa hanyoyin zahiri, sinadarai, da na halitta, tare da amfani da ƙarfi kamar wutar lantarki, sauti, haske, da maganadisu. Wannan labarin ya taƙaita amfani da "lantarki" a cikin fasahar sarrafa ruwa ta lantarki don magance wannan matsala.
Fasahar sarrafa ruwa ta lantarki tana nufin tsarin lalata gurɓatattun abubuwa a cikin ruwan shara ta hanyar takamaiman halayen lantarki, hanyoyin lantarki, ko hanyoyin zahiri a cikin wani takamaiman reactor na lantarki, ƙarƙashin tasirin electrodes ko filin lantarki da aka yi amfani da shi. Tsarin lantarki da kayan aiki suna da sauƙi, suna ɗaukar ƙaramin sawun ƙafa, suna da ƙarancin kuɗin aiki da kulawa, suna hana gurɓataccen abu na biyu yadda ya kamata, suna ba da babban ikon sarrafa halayen, kuma suna da amfani ga sarrafa kansa na masana'antu, wanda hakan ya sanya su laƙabi da fasahar "mai kyau ga muhalli".
Fasahar sarrafa ruwa ta lantarki ta haɗa da dabaru daban-daban kamar electrocoagulation-electroflotation, electrodialysis, electroadsorption, electro-Fenton, da kuma electrocatalytic advanced oxidation. Waɗannan dabarun sun bambanta kuma kowannensu yana da nasa aikace-aikace da fannoni masu dacewa.
Electrocoagulation-Electroflotation
A zahiri, electrocoagulation shine electroflotation, saboda tsarin coagulation yana faruwa a lokaci guda tare da flotation. Saboda haka, ana iya kiransa gaba ɗaya da "electrocoagulation-electroflotation."
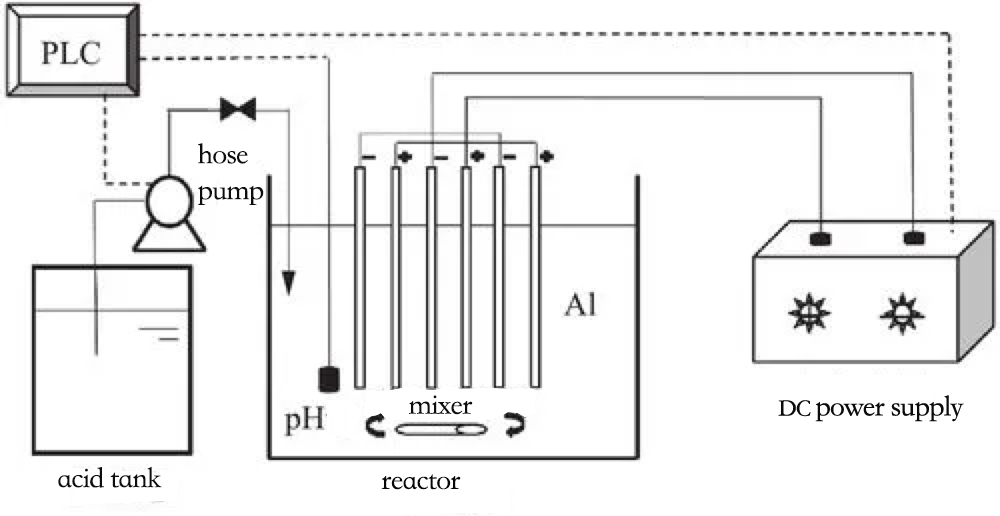
Wannan hanyar ta dogara ne akan amfani da wutar lantarki ta waje, wanda ke samar da cations masu narkewa a anode. Waɗannan cations suna da tasirin haɗuwa akan gurɓatattun abubuwa na colloidal. A lokaci guda, ana samar da isasshen iskar hydrogen a cathode ƙarƙashin tasirin wutar lantarki, wanda ke taimakawa kayan da aka yi amfani da su wajen tashi zuwa saman. Ta wannan hanyar, electrocoagulation yana cimma rabuwar gurɓatattun abubuwa da tsarkake ruwa ta hanyar anode coagulation da cathode flotation.
Ta amfani da ƙarfe a matsayin anode mai narkewa (yawanci aluminum ko iron), ions na Al3+ ko Fe3+ da aka samar yayin electrolysis suna aiki azaman coagulants na lantarki. Waɗannan coagulants suna aiki ta hanyar matse colloidal mai layi biyu, lalata shi, da kuma haɗa ƙwayoyin colloidal ta hanyar:
Al -3e→ Al3+ ko Fe -3e → Fe3+
Al3+ + 3H2O → Al(OH)3 + 3H+ ko 4Fe2+ + O2 + 2H2O → 4Fe3+ + 4OH-
A gefe guda, ana kiran sinadarin coagulant mai narkewa a cikin sinadarin M(OH)n a matsayin hadaddun sinadaran hydroxo masu narkewa kuma suna aiki a matsayin flocculant don haɗa su cikin sauri da inganci a cikin ruwan sharar gida yayin da suke haɗa su da haɗa su don samar da manyan tarawa, suna hanzarta tsarin rabuwa. A gefe guda kuma, ana matse colloids a ƙarƙashin tasirin electrolytes kamar aluminum ko iron gishiri, wanda ke haifar da coagulants ta hanyar tasirin Coulombic ko shaƙar coagulants.
Duk da cewa aikin electrochemical (rayuwar) na electroactive coagulants 'yan mintuna ne kawai, suna da tasiri sosai kan ƙarfin Layer biyu, don haka suna yin tasirin coagulation mai ƙarfi akan ƙwayoyin colloidal ko barbashi da aka dakatar. Sakamakon haka, ƙarfin shaye-shayen su da ayyukan su sun fi na hanyoyin sinadarai da suka haɗa da ƙara sinadaran gishirin aluminum, kuma suna buƙatar ƙananan adadi kuma suna da ƙarancin farashi. Electrocoagulation ba ya shafar yanayin muhalli, zafin ruwa, ko ƙazanta na halitta, kuma baya fuskantar halayen illa tare da gishirin aluminum da ruwa hydroxides. Saboda haka, yana da kewayon pH mai faɗi don magance ruwan sharar gida.
Bugu da ƙari, sakin ƙananan kumfa a saman cathode yana hanzarta karo da rabuwar colloids. Electro-oxidation kai tsaye akan saman anode da kuma electro-oxidation kai tsaye na Cl- zuwa chlorine mai aiki suna da ƙarfin oxidation mai ƙarfi akan abubuwan halitta masu narkewa da abubuwan da ba su da tsari a cikin ruwa. Sabbin hydrogen da aka samar daga cathode da oxygen daga anode suna da ƙarfin redox mai ƙarfi.
Sakamakon haka, hanyoyin sinadarai da ke faruwa a cikin na'urar lantarki suna da matuƙar rikitarwa. A cikin na'urar lantarki, hanyoyin lantarki, hanyoyin lantarki, da hanyoyin lantarki duk suna faruwa a lokaci guda, suna canza da kuma cire ƙwayoyin colloid da aka narkar da su da kuma gurɓatattun abubuwa da aka dakatar a cikin ruwa ta hanyar haɗakar ƙwayoyin halitta, flotation, da oxidation.
Xingtongli GKD45-2000CVC Wutar Lantarki ta DC
Siffofi:
1. Shigarwar AC 415V Mataki na 3
2. Sanyaya iska da aka tilasta
3. Tare da aikin haɓakawa
4. Tare da mitar awa mai amfani da amper da kuma na'urar watsa lokaci
5. Na'urar sarrafawa daga nesa tare da wayoyi masu sarrafa mita 20
Hotunan samfurin:


Lokacin Saƙo: Satumba-08-2023




